Pregnancy, COVID-19 and Emerging Therapeutic Options
Posted on: Tuesday 12 January 2021
Author: Allyah Abbas-Hanif, Sue Tansey, Sankarasubramanian Rajaram, Penny Ward
This article has been prepared by the following members of the Paediatrics and Pregnancy Expert Group of the Faculty of Pharmaceutical Medicine (FPM): Allyah Abbas-Hanif, Sue Tansey, Sankarasubramanian Rajaram and Penny Ward
It is provided for information and does not constitute advice or represent official FPM views or policy.
How to cite:
Abbas-Hanif, A et al. (2021), ‘Pregnancy, COVID-19 and Emerging Therapeutic Options’, Faculty of Pharmaceutical Medicine blog, 12 January 2021. Available at: <https://www.fpm.org.uk/blog/pregnancy-covid-19-and-emerging-therapeutic-options/> (Accessed: <DATE>).
Download this article as a PDF (210KB).

Introduction
Coronavirus-19 disease (COVID-19) continues to spread across the world, creating an unprecedented international public health crisis. With 131 million women giving birth annually, governments have taken steps to safeguard this group, earmarking them as at risk1. Although a low absolute risk of adverse outcomes have been reported, emerging data from the UK and US highlights an increased risk of pregnant women with COVID-19 requiring intensive care admission2,3,4. With new and faster spreading severe acute respiratory syndrome corona virus 2 (SARS-CoV-2) variants adding urgency to the vaccine push, we explore how the needs of pregnant women can be managed by the drug development community5. In this article we discuss the risks COVID-19 poses to pregnant women, the foetus and the newborn. We review current epidemiology, therapeutic options and focus on the specific drug development needs of this group during a pandemic.
Clinical manifestations of COVID-19 in pregnant women, the foetus and neonate
Women of reproductive age are reported to comprise 21% of all COVID-19 cases, 9% of whom were pregnant at the time6. Viral respiratory illnesses are associated with a higher risk of obstetric complications and adverse perinatal outcomes, including pneumonia, preterm labour and low birth weight7,8,9,10. The previous coronavirus infections: severe acute respiratory syndrome and Middle East respiratory syndrome demonstrated higher rates of maternal and foetal complications and mortality, warranting close attention to pregnant women in the current pandemic11. Data from prospective cohort studies suggested that maternal outcomes overall in COVID-19 were similar to non-pregnant 12,13. The incidence of hospitalisation of UK pregnant women with symptomatic SARS-CoV-2 has been estimated as 2.0 per 1000 maternities (March to 31 August 2020)2. The clinical situation is, however, evolving and results from a large and regularly updated US dataset suggest pregnant women with COVID-19 are at significantly higher risk of severe outcomes compared with non-pregnant women, including intensive care treatment, invasive ventilation, extracorporeal membrane oxygenation and death4. Although this retrospective study does have limitations, such as substantial missing data and pregnancy status ascertained in 36% of women of reproductive age only, it flags risks that cannot be ignored, particularly in the light of more infectious strains of SARS-CoV-2. Data from the UK has also recently indicated the proportion of pregnant women admitted to intensive care is increasing, especially in comparison to the first wave of COVID-193.
As with non-pregnant individuals, advanced age, obesity, pre-existing hypertension and diabetes are significant risk factors and increase the chances of a more severe disease presentation and evolution6,15. Interestingly, a recent living systemic review identified pregnant women as less likely to present with fever and myalgia15. This may be due to routine testing of all pregnant women and therefore the identification of asymptomatic sufferers.
SARS-CoV-2 infection may increase the risk of pre-term delivery to manage obstetric or medical complications, but rates of spontaneous pre-term labour do not appear to be elevated15. The incidence of stillbirth and neonatal death do not seem to be higher than the background rate2,4,15. Admission to neonatal units was noted to be increased in studies2. The rationale for this may include local policies on observation and quarantine of infants with exposure to SARS-CoV-2.
In utero vertical transmission has been reported in case studies and although rare, the SARS-CoV-2 genome has been found in umbilical cord blood, amniotic fluid, maternal vaginal mucosa and full term placenta16. Fortunately, infection of foetuses, newborns and infants is uncommon17. If neonates do become infected, most cases are asymptomatic or mild and outcomes are favourable18,19. Most complications in neonates born to COVID-19 positive mothers are a result of prematurity rather than COVID-19 infection20. Interestingly, both IgG and IgM antibodies against COVID-19 have been found in seronegative neonates born to COVID-19 infected mothers21. As IgM antibodies cannot cross the placenta, the suggestion of a foetal immune response against the virus is possible21.
Therapeutic options and specifics of drug development in pregnancy
Researchers around the world are working at record pace to progress treatment and prophylactic options for COVID-19. Any drug or vaccine candidate needs to be evaluated for risk/benefit to the pregnant women plus potential effects on the foetus, newborn and breastfed infant. As pregnant women are usually excluded from pre-registration clinical trials, other ways to accelerate the understanding of risk/benefit should be considered at the earliest opportunity and include the conduct of developmental and reproductive toxicity (DART) studies and using already established pregnancy and post-marketing registries22.
Vaccines
There are over 200 vaccines in development, with 63 vaccines in clinical stage evaluation23. A handful of vaccines are rolling out in select countries, including the novel mRNA immunisations by Pfizer-BioNTech and Moderna, plus AstraZeneca-University of Oxford’s viral vector vaccine. The phase 3 results from Janssen’s one dose vaccine are also expected shortly. These and other currently recruiting large scale vaccine studies have not actively recruited pregnant women, leading to limitations in safety data in a group that is potentially at risk.
Inclusion of pregnant women in vaccine trials, especially those looking to rapidly deliver results, is challenging but maternal immunisation is a successful tool and can, critically, provide dual protection. The influenza vaccine protects both the mother and infant and pertussis vaccines given in pregnancy afford passive protection to the infant24. The prospect of passive immunity through IgG transfer from a vaccinated pregnant mother is an attractive additional benefit for COVID-19 vaccines.
Phase 2 and 3 trials for vaccine studies include thousands of female subjects. Extrapolating data from women of childbearing age in clinical trials and those who inadvertently become pregnant offers invaluable insights. 49.4% of the Pfizer-BioNTech BNT162b2 Phase 3 trial were women and as of the 14th November 2020 datacut, 23 participants reported intercurrent pregnancy, outcomes of which are actively being followed25. DART studies can be conducted early in a clinical development plan and offer crucial safety insights of a drug’s effects on pregnancy and the foetus22. DART studies with BNT162b2 have revealed no vaccine-related effects on female fertility, pregnancy, or embryo-foetal development25. This data is also a useful safety indicator for other vaccines with similar mechanisms of action. The early completion of DART studies is critical and offers a catalytic step to earlier recruitment of pregnant women in trials of vaccines and novel agents.
The data on currently approved vaccines does not indicate any safety concerns, allowing the MHRA and FDA to recommend that clinicians undertake case by case assessment for the use of COVID-19 vaccines by pregnant women, particularly those with other high risk comorbidities26. SARS-CoV-2 variants have arisen in several locations including the UK and South Africa and although the current crop of vaccines are likely be effective for these, a Brazilian variant has been identified with changes to the receptor binding domain27,28. Drug developers from the 200 other vaccines in development should aim to evaluate safety markers in pregnancy and follow the successful precedent set by the approved vaccines to advance the inclusion of pregnant women in COVID-19 trials.
Medical treatments
Repurposing drugs with known safety profiles in pregnancy has been an attractive first approach with COVID-19. Remdesivir was used to treat pregnant women during the Ebola and Marburg virus epidemics29 and although the recent trial in COVID-19 did not include pregnant women, there were no significant safety concerns reported in women of childbearing potential30. Remdesivir has not shown a mortality benefit but has some clinical benefit in shortening the time to recovery, particularly in those requiring supplemental oxygen30. Remdesivir’s subsequent use by hospitalised pregnant women suffering with severe COVID-19 disease has occurred via expanded access programs31. Evaluation of the first 86 pregnant women to use the drug from 5 countries has been favourable, demonstrating high recovery rates within 28 days and low rates of serious adverse events31. The RECOVERY trial demonstrated a mortality benefit of low dose dexamethasone in patients with COVID-19 who required respiratory support32. This trial included pregnant and breastfeeding subjects and no pregnancy associated adverse outcomes were reported. The use of steroids and remdesivir are now included in national guidelines33. There are several trials looking at interferon (IFN) alpha and beta use in COVID-19, mostly in addition to antivirals. Results with injectable forms have been disappointing34. An investigational inhaled nebulised IFN beta-1a (SNG001) has however, shown some promise. When administered to hospitalised patients with COVID-19 in a phase 2b study, the likelihood of recovery by day 15 compared with placebo was increased34. Several studies (mostly in multiple sclerosis) have shown no increase in congenital abnormalities with INF use35, this coupled with the potentially to bypass the placenta makes nebulised IFN beta-1a an attractive option for pregnant women. DART studies should also be conducted with this investigational product to allow further insights to the risk/benefit in pregnant women. The use of other agents, such as monoclonal antibody therapies have shown some promise in clinical trials and case-reports of use in pregnant women are growing36,37. Given the potential for higher risk in pregnant women, in the absence of absolute contraindications, it may be reasonable to include pregnant women in clinical trials of these therapeutic approaches.
Conclusion
The race to find appropriate treatments and vaccines for COVID-19 is progressing swiftly, but with the urgency of more transmissible variants and more intensive care admissions, strategies to enrol pregnant women earlier into clinical development plans should be utilised. To ensure safety of pregnant women and neonates, drug developers should prioritise early initiation of DART studies followed by systematic review of inadvertent pregnancies during clinical trials in the general population. This, coupled with industry supported pregnancy registries and close collaboration with regulators and government bodies, will allow pregnant women to have access to investigational clinical trials whilst mitigating potential risks. The authors will continue to review developments for pregnant women during the COVID-19 pandemic.

References
- World Health Organization. Lerberghe, W. van and World Health Organization (2005) World Health Report 2005 make every mother and child count. https://www.who.int/whr/2005/en/ (accessed 10 Jan 2021).
- Vousden N, Bunch K et al. The incidence, characteristics and outcomes of pregnant women hospitalized with symptomatic and asymptomatic SARS-CoV-2 infection in the UK from March to September 2020: a national cohort study using the UK Obstetric Surveillance System (UKOSS). medRxiv 2021.01.04.21249195.
- Intensive care national audit & research centre (ICNARC). ICNARC report on COVID-19 536 in critical care: England, Wales and Northern Ireland, 8 January 2020. Napier House, 24 537 High Holborn, London: ICNARC,2020. https://www.icnarc.org/Our-Audit/Audits/Cmp/Reports (accessed 10 Jan 2021).
- Zambrano LD, Ellington S, Strid P, et al. Update: Characteristics of Symptomatic Women of Reproductive Age with Laboratory-Confirmed SARS-CoV-2 Infection by Pregnancy Status — United States, January 22–October 3, 2020. MMWR Morb Mortal Wkly Rep 2020;69:1641–1647.
- World Health Organizat SARS-CoV-2 Variants. Disease Outbreak News. https://www.who.int/csr/don/31-december-2020-sars-cov2-variants/en/ (accessed 10 Jan 2021).
- Ellington S, Strid P, et al. Characteristics of women of reproductive age with laboratory-confirmed sars-cov-2 infection by pregnancy status – United States, January 22-June 7, 2020. MMWR Morb Mortal Wkly Rep. 2020;69:769-75.
- Chen YH, Keller J, et al. Pneumonia and pregnancy outcomes: a nationwide population-based study. Am J Obstet Gynecol. 2012 Oct;207(4):288.e1-7.
- Ramsey PS, Ramin KD. Pneumonia in pregnancy. Obstet Gynecol Clin North Am. 2001;28:553–569.
- Siston AM, Rasmussen SA et al. Pandemic 2009 influenza A (H1N1) virus illness among pregnant women in the United States. JAMA. 2010;303:1517–1525.
- Banhidy F, Acs N et al. Maternal acute respiratory infectious diseases during pregnancy and birth outcomes. Eur J Epidemiol. 2008;23:29– 35.
- Mullins E, Evans D, et al. Coronavirus in pregnancy and delivery: rapid review. Ultrasound Obs Gynecol. 2020;55:586-592.
- Adhikari EH, Moreno W, et al. Pregnancy outcomes among women with and without Severe Acute Respiratory Syndrome Coronavirus 2 infection. JAMA Netw Open. 2020;3(11):e2029256.
- Knight M, Bunch K, et al. Characteristics and outcomes of pregnant women hospitalised with confirmed SARS-CoV-2 infection in the UK: a national cohort study using the UK Obstetric Surveillance System (UKOSS). BMJ 2020;369:m2107.
- Fogacci S, Federica F. Management of pregnancy-related hypertensive disorders in patients infected with SARS CoV-2: pharmacological and clinical issues, EHJ – CV pharma. pvaa105.
- Allotey J, Stallings E, et al. Clinical manifestations, risk factors, and maternal and perinatal outcomes of coronavirus disease 2019 in pregnancy: living systematic review and meta-analysis. BMJ.2020;370:m3320.
- Fenizia, C., Biasin, et al. Analysis of SARS-CoV-2 vertical transmission during pregnancy. Nat Commun 11, 5128 (2020).
- Di Mascio D, Khalil A, et al. Outcome of coronavirus spectrum infectons (SARS, MERS, COVID-19) during pregnancy: a systematic review and meta-analysis. Am J Obstet Gynecol . 2020 May;2(2):100107.
- Zeng, L. et al. Neonatal early-onset infection with SARS-CoV-2 in 33 neonates born to mothers with COVID-19 in Wuhan, China. JAMA Pediatr. 174, 722–725 (2020).
- Chen H, Guo J, et al. Clinical characteristics and intrauterine vertical transmission potential of COVID-19 infection in nine pregnant women: a retrospective review of medical records. Lancet. 2020;395:809-81.
- Flaherman VJ, Afshar Y, et al. Infant outcomes following maternal infection with SARS-CoV-2: first report from the PRIORITY study. Clin Infect Dis. ciaa1411.
- Carosso A, Cosma S et al. How to reduce the potential risk of vertical transmission of SARS-CoV-2 during vaginal delivery. Eur J Obstet Gynecol Reprod Biol. 2020;250:246-249.
- ICH guideline M3(R2) Guidance on nonclinical safety studies for the conduct of human clinical trials and marketing authorization for pharmaceuticals. https://database.ich.org/sites/default/files/M3_R2__Guideline.pdf (accessed 10 Jan 2021).
- World Health Organization Draft landscape of COVID-19 candidate vaccines. https://www.who.int/publicatons/m/item/draft-landscape-of-covid-19- candidate-vaccines (accessed 10 Jan 2021).
- Shakib JH, Korgenski K, et al. Influenza in Infants Born to Women Vaccinated During Pregnancy. Pediatrics. 2016 Jun;137(6):e20152360.
- Pfizer-BioNTech’s BNT162 VRBPAC Briefing document https://www.fda.gov/media/144246/download (accessed 10 Jan 2021).
- Royal College of Obstetricians and Gynaecologists. Updated advice on COVID-19 vaccination in pregnancy and women who are breastfeeding. https://www.rcog.org.uk/en/news/updated-advice-on-covid-19-vaccination-in-pregnancy-and-women-who-are-breastfeeding/ (accessed 10 Jan 2021).
- Voloch CM, da Silva R, et al. Genomic characterization of a novel SARS-CoV-2 lineage from Rio de Janeiro, Brazil. medRxiv 2020.12.23.20248598;(accessed 10 Jan 2021).
- Resende PC, Bezerra JF, et al.Spike E484K mutaton in the first SARS-CoV-2 reinfecton case confirmed in Brazil. Case report. 2020. https://virological.org/t/spike-e484k-mutation-in-the-first-sars-cov-2-reinfection-case-confirmed-in-brazil-2020/584 (accessed 10 Jan 2021).
- Mulangu S, Dodd LE, et al. A randomized, controlled trial of Ebola virus disease therapeuses. N Engl J Med. 2019; 381: 2293-2303.
- Beigel JH, Tomashek KM, et al. Remdesivir for the treatment of Covid- 19 — final report. N Engl J Med 2020;383:1813-1826.
- Burwick RM, Yawetz S et al. Compassionate Use of Remdesivir in Pregnant Women With Severe Coronavirus Disease 2019, Clin Infect D, ciaa146.
- RECOVERY Collaborative Group. Effect of Dexamethasone in Hospitalized Patients with COVID-19 – Preliminary Report Group. N Engl J Med. 2020 Jul 17: NEJMoa2021436.
- Royal College of Obstetricians and Gynaecologists. Coronavirus (COVID-19) infection in pregnancy. RCOG guideline; London (UK): RCOG; 2020. https://www.rcog.org.uk/globalassets/documents/guidelines/2020-10-14-coronavirus-covid-19-infection-in-pregnancy-v12.pdf, (accessed 10 Jan 2021).
- Monk PD, Marsden RJ, et al. Safety and efficacy of inhaled nebulised interferon beta-1a (SNG001) for treatment of SARS-CoV-2 infection: a randomised, double-blind, placebo-controlled, phase 2 trial. Lancet Respir Med 2020 Nov 12:S2213-2600(20)30511-7.
- Hellwig K, Geissbuehler Y, et al. Pregnancy outcomes in interferon-beta-exposed patients with multiple sclerosis: results from the European Interferon-beta Pregnancy Registry. J Neurol. 2020 Jun;267(6):1715-1723.
- REMAP-CAP Investigators, Gordon AC. Interleukin-6 receptor antagonists in critically ill patients with covid-19—preliminary report. MedRxiv 2021.01.07.21249390.
- Naqvi M, Zakowski P,et al. Tocilizumab and Remdesivir in a Pregnant Patient With Coronavirus Disease 2019 (COVID-19). Obstet Gynecol. 2020 Nov;136(5):1025-1029.
Read our COVID-19 blog articles

It’s All Greek to Me: SARS-CoV-2 Variants, Vaccines and Antivirals
A Deep Dive by Professor Penny Ward et al
FPM Statement on COVID Vaccine Supply May 2021
FPM urges governments, industry and regulators to collaborate to find solutions to ensure supply as soon as soon as possible.
Inhalation therapies for COVID-19
Can we inhibit the initial stages of the infection of the respiratory tract?

Pregnancy, COVID-19 and Emerging Therapeutic Options
Allyah Abbas-Hanif, Sue Tansey, Sankarasubramanian Rajaram, Penny Ward

FPM Clinical Trials Resilience Survey – Report Published
Adaptations and innovations made to clinical trial programmes and regulatory frameworks during the COVID-19 pandemic.

Blog article: Preparedness Planning on Public Health Emergencies (PHEs)
The webinar series that has brought FPM into the heart of the COVID-19 pandemic.
What you need to know about COVID-19 testing
An article by Robert L Holland FFPM and John Bagshaw of the British In Vitro Diagnostics Association

Press release: Faculty of Pharmaceutical Medicine launches ‘Principles for Clinical Studies into COVID-19’ guidance
Read the press release

COVID-19 – how pharmaceutical medicine is meeting the challenge
A report of the FPM survey of members on the effect of COVID-19 on their professional lives
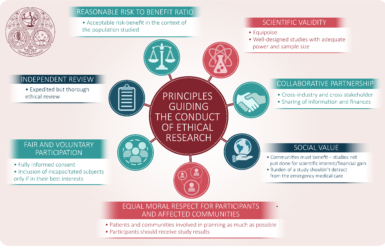
The ethics of conducting clinical trials in the search for treatments and vaccines against COVID-19
Some of the important ethical issues when planning and conducting clinical trials in the COVID 19 pandemic.
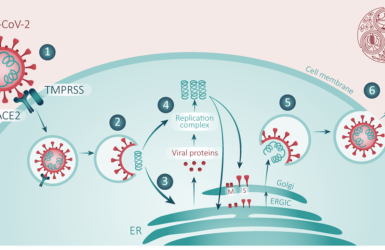
COVID-19/SARS-CoV-2 Pandemic
A landmark review by Dr Penny Ward et al addressing current knowledge concerning SARS-CoV-2 and COVID-19 disease.