It’s All Greek to Me: SARS-CoV-2 Variants, Vaccines and Antivirals
Posted on: Thursday 23 December 2021
Author: Penny Ward et al
This FPM Deep Dive article has been prepared by Professor Penny Ward with input from FPM’s COVID-19 Team, notably Dr Renata Crome, Dr Flic Gabbay, and Dr Sheuli Porkess.
It is provided for information and does not constitute advice or represent official FPM views or policy.
How to cite:
Ward, P et al (2021), ‘It’s All Greek to Me: SARS- CoV-2 Variants, Vaccines and Antivirals’, FPM Deep Dive, 23 December 2021. Available at: https://www.fpm.org.uk/deep-dive/greek-to-me/ (Accessed: <date>).

Introduction
The recent emergence and rapid spread of the Omicron variant of the SARS-CoV-2 virus has certainly set the cat among the pigeons. Following the successful roll out of the COVID-19 vaccines across the UK adult and adolescent populations, the arrival of a new, apparently less vaccine susceptible, variant threatens to undo all the good work.
So, what do we know about the various COVID-19 viral variants associated with the various epidemics of illness in the UK? What is different about the Omicron variant and what can be done to ameliorate its potential impact over the winter?

Viral Variation
A common feature of RNA viruses is their ability to tolerate significant mutability in replicating their genome while retaining ‘fitness’ – the ability to replicate within and transmit infection between affected hosts. A progeny virus with one or more mutations is called a “variant” of the original. To identify the potential for emergent viral variants of SARS-CoV-2, the genetic material of viruses isolated from affected cases is sequenced and the derived sequence compared to the original Wuhan strain and to other previously identified variants to determine if a new strain has emerged. Sequenced genomes are made publicly available via a number of data repositories, including the Global Initiative on Sharing Avian Influenza Data (GISAID)1 platform curated by the World Health Organisation (WHO) and/or Nextstrain2. In each case, viral strains are cross compared to produce a viral phylogenetic ‘tree’ facilitating the tracking of the evolution of strains identified around the world. Several nomenclature systems are in use, including the viral ‘clade’ systems derived from GISAID and Nextstrain, and also from an algorithm proposed by Rambaut and colleagues known as the Pango nomenclature3,4. Each system provides a unique identifier for each strain, derived from the ‘parent’ strain to which it is most closely similar but differ in the number of distinct variants that may be identified within each system; by July 2021 there were 1,293 Pango lineages identified, compared to 12 and 9 clades for the NextStrain and GISAID nomenclatures, respectively. SARS-CoV-2 has to date given rise to multiple variants with distinct mutation profiles in multiple areas of the world, with many millions of unique sequences held within the sequence data repositories.
Variants identified are further classified according to their biologic properties, as proposed by the WHO:
A SARS-CoV-2 variant of interest (VOI) is a SARS-CoV-2 variant:
- with genetic changes that are predicted or known to affect virus characteristics such as transmissibility, disease severity, immune escape, diagnostic or therapeutic escape; AND
- that has been identified as causing significant community transmission or multiple COVID-19 clusters, in multiple countries with increasing relative prevalence alongside increasing number of cases over time, or other apparent epidemiological impacts to suggest an emerging risk to global public health.
A SARS-CoV-2 Variant of Concern (VOC) is a SARS-CoV-2 variant that meets the definition of a VOI (see above) and, through a comparative assessment, has been demonstrated to be associated with one or more of the following changes at a degree of global public health significance:
- increase in transmissibility or detrimental change in COVID-19 epidemiology; OR
- increase in virulence or change in clinical disease presentation; OR
- decrease in effectiveness of public health and social measures or available diagnostics, vaccines, therapeutics
Variants that are identified but about which little is known are termed variants under monitoring (VUM), pending their biologic properties being better understood.
In 2021 WHO convened a group of scientists from the WHO Virus Evolution Working Group (now called the Technical Advisory Group on Virus Evolution), the WHO COVID-19 reference laboratory network, representatives from GISAID, Nextstrain, Pango and additional experts in virological, microbial nomenclature and communication from several countries and agencies to consider easy-to-pronounce and non-stigmatising labels to facilitate information sharing for VOI and VOC. The group recommended using letters of the Greek Alphabet, i.e., Alpha, Beta, Gamma, Delta etc as this was considered to be an easier and more practical system for use in discussion with non-scientific audiences. Letters which might cause confusion are not used, which is why the letters Nu (sounds like new) and Xi (a Chinese surname) have not been assigned to any SARS-CoV-2 variant strain.
Current Variants of Concern
Table 1 identifies Variants of Concern (VOCs) which have been identified to date5.
| WHO label | Pango lineage | GISAID clade | Nextstrain clade | Additional amino acid changes monitored | Earliest documented samples | Date of designation |
|---|---|---|---|---|---|---|
| Alpha | B.1.1.7 | GRY | 20I (V1) | +S:484K +S:452R | United Kingdom, September 2020 | 18 December 2020 |
| Beta | B.1.351 | GH/501Y.V2 | 20H (V2) | +S:L18F | South Africa, May 2020 | 18 December 2020 |
| Gamma | P.1 | GR/501Y.V3 | 20J (V3) | +S:681H | Brazil, November 2020 | 11 January 2021 |
| Delta | B.1.617.2 | G/478K.V1 | 21A, 21I, 21J | +S:417N +S:484K | India, October 2020 | VOI: 4 April 2021 | VOC: 11 May 2021 |
| Omicron | B.1.1.529 | GRA | 21K, 21L | +R346K | Multiple countries, November 2021 | VUM: 24 November 2021 | VOC: 26-Nov 2021 |
Here in the UK, the Alpha variant was the dominant strain type during the second wave in December 2020, the Delta variant has dominated the third wave which started in June 2021 and the Omicron variant, first identified in November, is rapidly increasing and, as we near the end of 2021, may in some locations be overtaking the Delta variant as the dominant strain. The Omicron strain contains over 60 mutations in multiple areas of the spike protein, the target antigen for all current vaccines and some COVID therapeutic antibodies, as well as in other areas of the viral genome which may result in altered biological properties relative to the Delta variant which has dominated the UK epidemic during 2021.
The Omicron Variant
The mutations identified in this variant include the following genetic changes in the spike protein, the multidomain open reading frame region coding various non structural proteins (ORF1ab}, the envelope protein, the membrane protein and the nucleocapsid protein as follows:
- Spike protein: A67V, Δ69-70, T95I, G142D, Δ143-145, Δ211, L212I, ins214EPE, G339D, S371L, S373P, S375F, K417N, N440K, G446S, S477N, T478K, E484A, Q493R, G496S, Q498R, N501Y, Y505H, T547K, D614G, H655Y, N679K, P681H, N764K, D796Y, N856K, Q954H, N969K, L981FHalf (15) of these 30 changes are located in the receptor binding domain-RBD (residues 319–541) which suggest that the strain could be less sensitive to antibodies induced by vaccines derived from the original SARS-CoV-2 strain. In addition, monoclonal antibody therapeutics targeting this region of the spike protein may be less likely to neutralise the strain.
- ORF1ab
- nsp3: K38R, V1069I, Δ1265, L1266I, A1892T
- nsp4: T492I
- nsp5: P132H
- nsp6: Δ105-107, A189V
- nsp12: P323L
- nsp14: I42V
- Envelope protein: T9I
- Membrane protein: D3G, Q19E, A63T
- Nucleocapsid protein: P13L, Δ31-33, R203K, G204R
Clinical Epidemiology of Disease Caused by the Omicron Variant
Initial data from South Africa suggest that this variant spreads rapidly within affected communities and is capable of reinfecting individuals that had been fully vaccinated or that had previously recovered from COVID-19. Although symptoms observed among cases caused by the Omicron variant were reportedly mild, cold like symptoms, hospitalisation rates have been increasing in affected provinces, particularly among younger children. In an early report the South African Medical Research Council (SAMRC) suggested that the majority of hospitalised patients did not require oxygen supplementation and few needed ICU admission6. However, as more severe complicated illness may take time to emerge and the majority of the patients admitted in South Africa were younger age adults it is still too early to completely understand the clinical epidemiological picture of disease caused by this strain and particularly to understand potential impact in populations with a higher proportion of older age adults.
Vaccine Effectiveness vs the Omicron Variant
Early data investigating the neutralising effect of post vaccination sera on the Omicron variant in vitro suggest a potential reduction of vaccine effectiveness with neutralising antibody titres being significantly lower than against previously circulating variants, including the Delta variant7,8,9. An early assessment of the clinical effectiveness of vaccines deployed in the UK suggested complete loss of protection against symptomatic disease by week 15 following completion of a primary course of the ChAdOx01 vaccine and ~30-40% at the same timepoint following receipt of the BNT162b vaccine. Protection against symptomatic disease caused by the Omicron strain increased to 70-80% following receipt of a BNT162b booster shot regardless of the primary course type (Figure 1). The authors comment that protection against severe disease could not be investigated in this study due to the small number of cases, younger age and short period of follow up available for affected cases at the time of investigation10.
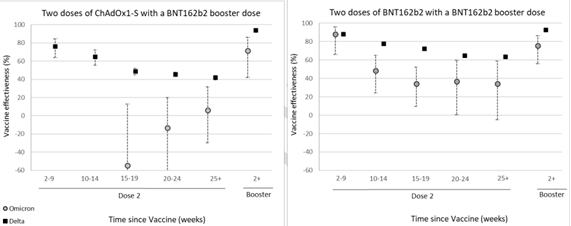
An epidemic model projection for the UK has estimated that in the face of reduced vaccine effectiveness, increased transmission rate and assuming a disease severity pattern similar to that caused by the Delta variant, significant healthcare service burden could emerge with peak case load and hospitalisations similar to those seen during the winter 2020-21 in England unless non pharmaceutical interventions are introduced rapidly11. Hogan et al, using a model which incorporates the immunological response to vaccination into estimates of vaccine protection against severe disease suggested that protection against severe disease could fall to 80.1% (95% CrI 76.3%–83.2) by 60 days post boost if neutralising antibody levels decay at the same rate following boosting as following the primary course12. If neutralising antibody levels decay more slowly, protection will be sustained at a higher level. It is too early to estimate the duration of protection offered by booster shots at this time, but if antibody decay post booster follows the same trajectory as following the primary course, revaccination at 2-3 month intervals may be needed to sustain high level protection against severe disease caused by this variant.
mRNA and viral vector vaccine technologies can be rapidly amended to include new strains with significantly reduced susceptibility to current vaccine strains. In addition, the use of in vitro approaches to enable an understanding of the level of antibody needed to neutralise the new strain following human administration can enable the use of immunobridging approaches to predict potential clinical effectiveness of a new vaccine negating the need for large clinical effectiveness trials. This approach has been used for the control of influenza for the past 80 years and is currently undergoing some adaptation to enable application for COVID vaccines13.
In meantime, in the face of a degree of uncertainty on the clinical effectiveness of current vaccines and the potential need to revaccinate at frequent intervals, even if a revised vaccine is developed, interest has turned to the potential deployment of antiviral therapeutics to further mitigate disease severity and relieve pressure on healthcare services.
Therapeutics
Over the past year, a number of monoclonal antibody therapeutics and two oral antiviral agents have been shown to reduce hospitalisation and mortality among high-risk patients with COVID-19, provided that treatment was given early (within five days) of first symptom onset. In addition, monoclonal antibody therapy was shown to protect close contacts of COVID-19 affected patients against infection and illness [post exposure prophylaxis] and to provide pre-exposure protection against infection and illness during an ongoing community epidemic.
Monoclonal Antibody Therapy
Five companies have developed monoclonal antibody treatments for COVID-19 and completed phase III studies. Three of these products are monoclonal antibodies (MAbs) or combination of MAbs directed against the receptor binding domain (RBD) of the spike protein of the SARS-CoV-2 virus. This is a highly mutable portion of the spike protein and, perhaps unsurprisingly, these three agents, the combination of bamlanivimab and etesevimab (Eli Lilly), the single treatment regdanvimab (Regkirona, Celltrion) and the combination of casirivimab and imdevimab (Ronapreve, Regeneron/Roche) are not effective in vitro against the Omicron variant14. These agents remain potentially effective against illness caused by other variants based on the results of Phase III trials conducted during outbreaks of illness caused by other variants15,16,17.
| Antibody | Population | Treatment Window | Endpoint | Treatment n/N (%) | Placebo n/N (%) | Effect |
|---|---|---|---|---|---|---|
| Bamlanivimab and etesevimab (Eli Lilly) | Unvaccinated high risk adults and adolescents >12 years | Up to 10 days post onset of symptoms | Proportion of patients hospitalised/dying within 28 days | 11/518 (2%) | 36/517 (7%) | 5% absolute 70% relative reduced |
| Regdanvimab (Regkirona, Celltrion) | Unvaccinated high risk adults >18 years | Up to 7 days post onset of symptoms | Proportion of patients progressing to severe disease, hospitalisation or dying within 29 days | 14/446 (3.1%) | 48/434 (11.1%) | 8% absolute,73% relative reduced |
| Casirivimab and Imdevimab (Ronapreve, Regeneron/Roche | Unvaccinated high risk adults and adolescents >12 years | Up to 7 days post onset of symptoms | Proportion of subjects with ≥ 1 COVID-19-related hospitalisation or all-cause death within 29 days | 11/1192 (0.9%) | 40/1193 (3.4%) | 2.5% absolute72.5% relative reduced |
Two other antibodies treatments, sotrovimab (Xevudy, GSK/Vir) and the combination of tixagevimab and cilgavimab (Evusheld, AstraZeneca) are antibodies directed against highly conserved portions of the SARS-CoV-2 spike protein. As the binding target domains are in conserved regions, these agents may be less susceptible to spike protein mutations. Indeed, both are reported to have sustained activity against the Omicron variant in vitro and might be expected to retain similar effectiveness to the outcomes described in completed Phase III trials. At the time of writing only data for sotrovimab has been reported in full (Table 3)18.
| Antibody | Population | Treatment Window | Endpoint | Treatment n/N (%) | Placebo n/N (%) | Effect |
|---|---|---|---|---|---|---|
| Sotrovimab (Xevudy, GSK/Vir) | Unvaccinated high risk adults | Up to 5 days post onset of symptoms | Proportion of patients requiring hospitalisation/dying within 28 days | 6/528 (1%) | 30/529 (6%) | 5% absolute 79% relative reduced |
The safety profile of all of these products was limited to occasional infusion reactions which were rarely serious. Because the antibodies are protein biologicals, no drug interactions are anticipated. Although not studied in pregnant women, these agents could be used in pregnancy if needed, to prevent more serious disease.
Prevention Studies
These same monoclonal antibody treatments can also be used as ‘passive prevention’ approaches to prevent coronavirus infection and illness in individuals exposed to potential for infection. Two approaches can be taken: immediate intervention to prevent illness developing among close contacts of an identified case (post exposure prevention/prophylaxis) or pre-exposure prevention for individuals living and going about their usual business within the community at the time of a COVID outbreak. Post exposure prevention/prophylaxis is useful to prevent sustained outbreaks occurring in ‘closed’ institutions for example care homes or prisons, and may make the more efficient use of restricted supply. Pre-exposure prevention might be considered as an add on to or replacement for vaccination in high risk individuals who may not have responded adequately to vaccines. As potential exposure to infection may occur over a prolonged period, depending on the duration of a community epidemic, this approach may result in high drug usage within the population, many of whom may not be at significant risk of exposure to an infected case if the community attack rate is low. Bamlanivimab, the casirivimab and imdevimab combination and the tixagevimab and cilgavimab combination MAbs products have all been investigated in prevention trials19,20,21. Bamlanivimab was investigated in a randomized, double-blind, single-dose, phase 3 trial that enrolled residents and staff of 74 skilled nursing and assisted living facilities in the United States with at least 1 confirmed SARS-CoV-2 index case. A total of 1175 participants enrolled in the study from August 2 to November 20, 2020, when the alpha variant was the dominant strain in circulation in the USA. The trial population comprised a total of 966 participants (666 staff and 300 residents) who were negative at baseline for SARS-CoV-2 infection and serology. Bamlanivimab significantly reduced the incidence of COVID-19 in the population compared with placebo (8.5% vs 15.2%; odds ratio, 0.43 [95% CI, 0.28-0.68]; P < .001. Five deaths attributed to COVID-19 were reported by day 57; all occurred in the placebo group. Efficacy was most marked in the elderly resident population in this study than among the care home staff. In the resident prevention population, incidence of mild or worse COVID-19 was significantly lower in the bamlanivimab group compared with the placebo group (8.8% vs 22.5%; odds ratio, 0.20; 95% CI, 0.08-0.49; P < .001)19. O’Brien and colleagues investigated the casirivimab and imdevimab combination product in a household setting. In this study, participants (≥12 years of age) were enrolled and randomised 1:1 within 96 hours after a household contact received a diagnosis of SARS-CoV-2 infection to receive a total dose of 1200 mg of the MAb or matching placebo administered by subcutaneous injection. The trial recruited patients from July 2020, when the initial Wuhan strain and later the alpha variant were the predominant strains in the US. Participants were stratified according to the results of the local diagnostic assay for SARS-CoV-2 and according to age. The primary efficacy end point was the development of symptomatic SARS-CoV-2 infection through day 28 in participants who did not have SARS-CoV-2 infection (as measured by reverse-transcriptase-quantitative polymerase-chain-reaction assay) or previous immunity (seronegativity). Symptomatic SARS-CoV-2 infection developed in 11 of 753 participants in the MAb group (1.5%) and in 59 of 752 participants in the placebo group (7.8%) (relative risk reduction [1 minus the relative risk], 81.4%; P<0.001). In weeks 2 to 4, a total of 2 of 753 participants in the MAb group (0.3%) and 27 of 752 participants in the placebo group (3.6%) had symptomatic SARS-CoV-2 infection (relative risk reduction, 92.6%). The MAb treatment also prevented symptomatic and asymptomatic infections overall (relative risk reduction, 66.4%) and shortened the duration of illness from 3.2 to 1.2 weeks. In addition, the duration of a high viral load (>104 copies per milliliter) was also shorter (0.4 weeks and 1.3 weeks, respectively in MAb and placebo recipients)20. Lastly, the tixagevimab and cilgavimab combination MAb has recently received emergency use authorisation in the USA, based on results from two prevention trials, one (PROVENT) looking at pre-exposure prevention and the other (STORMCHASER) looking at post exposure prevention in a household setting. This product is administered as a single IM injection and, possibly because IM injection is not as efficient as IV injection in rapidly achieving adequate plasma concentrations, the post exposure prevention trial did not succeed in demonstrating prevention of illness within the first two weeks after administration in close contacts of COVID patients. However, the pre-exposure trial and the longer term follow-up of the post exposure prevention study both demonstrated that the single administration of this MAb combination prevented the development of symptomatic disease by ~77% relative to placebo when given prior to exposure to a known case of COVID21.
Antiviral Therapy
Direct acting antiviral treatment is an alternative approach for the management of SARS-CoV-2 variant mediated disease. It is more difficult for viruses to change the structure and function of viral enzymes which contribute to viral replication in vivo than to change the antigenic structure of its coat and so antiviral medicines targeted against these proteins may retain activity against new variants and enable treatment options for affected patients. Three antiviral treatments are either approved in the UK or under review and thought to be likely to be approaching approval for the treatment of COVID.
Remdesivir: Three Day Treatment in High Risk Outpatients
The first of these, remdesivir, is a nucleoside analogue which inhibits the RNA polymerase of the SARS-CoV-2 virus. It was approved for UK use in hospitalised patients in 2020 following the outcome of the ACTT-1 study22, which demonstrated faster recovery from COVID among remdesivir treated patients than those given placebo. It has to be given intravenously once daily for 5 days (or up to 10 days if required) and because of the route of administration has been considered impractical for outpatient use. A Phase 3 randomized, double-blind, placebo-controlled trial (PINETREE) was conducted to evaluate the efficacy and safety of a three-day course of IV remdesivir, started within seven days of first onset of symptoms, for the treatment of COVID-19 in non-hospitalised patients at high risk for disease progression. In an analysis of 562 participants randomly assigned in a 1:1 ratio to receive remdesivir or placebo, remdesivir demonstrated a statistically significant, 87% reduction in risk for the composite primary endpoint of COVID-19 related hospitalization or all-cause death by Day 28 (0.7% [2/279]) compared with placebo (5.3% [15/283]) p=0.008 23. The EMA has extended the approval of remdesivir in the EU to permit outpatient use of this treatment, based on the result of this trial24. The shorter course of treatment required may enable the use of the treatment in an outpatient setting to reduce the burden on hospital care in the face of a new emergent wave caused by the Omicron variant. Gilead has confirmed that remdesivir has sustained antiviral activity against all major previously identified variants of SARS-CoV-2 including Alpha, Beta, Gamma, Delta, and Epsilon. Due to the similarities in the viral RNA polymerase, these laboratory findings suggest that remdesivir will also continue to be active against the Omicron variant25. As a result, although the PINETREE trial was conducted during the US alpha wave in early 2021, the results should be applicable to disease caused by the Omicron variant.
Molnupiravir
The UK was the first country in the world to approve the use of molnupiravir for the treatment of COVID. This agent was approved in the UK on the basis of an interim analysis of a trial in unvaccinated patients with at least one risk factor (Age >60 years, Active Cancer, Chronic Kidney Disease, Chronic Obstructive Pulmonary Disease, Obesity (BMI ≥ 30), Serious Heart Condition, Diabetes Mellitus) increasing the potential need for hospitalisation that presented within 5 days of first onset of symptoms of illness. In an interim analysis which included data from 775 randomised participants, 1600mg daily Molnupiravir, given as 4x200mg capsules twice daily, reduced the risk of hospitalisation or death by approximately 50%; 7.3% of participants who received molnupiravir were either hospitalised or died through Day 29 following randomisation (28/385), compared with 14.1% of placebo-treated participants (53/377); p=0.0012. Clinical data are now available for all 1433 randomised patients. In the final analysis including all randomised patients, molnupiravir reduced the risk of hospitalisation or death by approximately 30% (from 9.7% (68/699) among placebo recipients to 6.8% (48/709) among patients taking molnupiravir)26. Because of this apparent reduction in effectiveness in the final analysis of the trial in unvaccinated patients and also to investigate the effectiveness of the agent in patients that have already been vaccinated, the UK is conducting a second trial, the PANORAMIC study, to provide further information in both unvaccinated and vaccinated patients who may be at higher risk of needing hospital treatment, should they develop COVID. Safety concerns with this product include the potential for emergence of viral mutations due to its mechanism of action, teratogenicity and genotoxicity. Molnupiravir is not recommended for use by pregnant women. With the exception of these concerns, molnupiravir was reported to be well tolerated in the phase III trial, with minor GI effects being the most frequently reported adverse effects.
Nirmatrelvir
Nirmatrelvir is the recently announced INN for PF-07321332, a specifically designed inhibitor of the SARS-CoV-2 main protease which has been in clinical trials for the treatment of COVID since 202027. After early dose range finding studies, a phase III daily dose of 600mg nirmatrelvir given as 2x 150mg tablets twice daily together with ritonavir 100mg ( a CYP3A4 inhibitor, which enables antiviral concentrations of nirmatrelvir to be sustained in the body over the ~12 hour dose interval), has been investigated in COVID-19 affected high risk unvaccinated patients in one study (EPIC-HR) and in a second study which included patients considered to be at lower risk some of whom had been previously vaccinated (EPIC-SR). Results from these studies have been made public in a press release but have yet to be published. Based on the data from the press release, treatment with nirmatrelvir/ritonavir reduces the risk of hospitalization or death for any cause by 88% compared to placebo in patients treated within five days of symptom onset; 0.8% of patients who received nirmatrelvir/ritonavir were hospitalized or died through Day 28 following randomization (8/1039 hospitalized with no deaths), compared to 6.3% of patients who received placebo (66/1046 hospitalized with 12 subsequent deaths) (p<0.0001). Treatment within 3 days of symptom onset was slightly more effective (89% relative reduction compared to placebo). The standard risk patient study used a novel endpoint to evaluate time to sustained recovery, which was missed in an interim analysis. However, consistent with EPIC-HR, early treatment in the EPIC-SR study reduced the proportion of patients needing hospital care from 2.4 to 0.7% in the placebo and active treatment groups respectively, overall a 70% reduction in this outcome among these lower risk patients. Pfizer has also announced a study investigating the product for post exposure prevention which began in September this year. The EMA has released advice to EU member states considering early use of this agent while it remains under regulatory review in mid December28. Because of the co administration of this product with the potent CYP3A4 inhibitor ritonavir, use with a range of medicines commonly taken by older patients for other comorbidities is not recommended. This may restrict exploitation of this product in older, frailer patients requiring multiple concomitant treatments for other disorders. Although the product is reported to be well tolerated, complete safety information has yet to be published.
Resistance to Monoclonal Antibody Treatments and Antivirals: The need for monitoring
The principal concern with the use of specific treatment with antiviral therapy is the potential for emergent resistance. This is particularly likely to emerge in patients with immune suppression, a group at particular risk from COVID-19. Although antiviral treatment can reduce the quantity of virus in an affected patient, efficient clearance depends on the immune system and immune suppressed patients may continue to replicate virus at low level while being treated, increasing the risk of selection of strains resistant to the treatment given. In vitro resistance studies have shown (as they are designed to) potential emergence of mutated viruses with reduced sensitivity to molnupiravir and to the monoclonal antibody treatments, although clinically important resistance to these agents has not been reported during clinical use to date. Emergence of remdesivir resistant virus has been observed clinically, but may be overcome by repeated courses of treatment or addition of antibody therapy in affected patients29,30,31.
As these agents are pressed into potential widescale use, systems will need to be in place to regularly sample treated patients to detect potential for emergent resistant strains as rapidly as possible. In addition, consideration should be given to the use of combination therapy in more severely immune-suppressed patients to reduce the potential for antiviral drug resistance to the few antivirals we have currently available, as discussed in a NERVTAG briefing paper published in December 202232. The influenza antiviral susceptibility testing framework offers a potential model for such a surveillance system33.
Conclusions
The ongoing COVID-19 pandemic has been characterised by the emergence and spread of multiple variants. The arrival of a novel variant with reduced sensitivity to current vaccines represents a significant threat to the progress made so far in controlling this disease and reducing its impact. While rapid amendment of vaccine strains can be attempted, unless the apparent necessity to readminister vaccines at 2-3 monthly intervals is overcome, vaccination may become impractical for the longer term management of this condition. This suggests that greater attention needs to be paid to the rapid discovery, development and deployment of antiviral treatments to reduce disease severity and offer alternative methods for outbreak control. While the products we have currently available may be useful current supply limitations require difficult decisions to be made on availability of treatment within the population. A larger stockpile of treatments will be needed for future proofing than have currently been envisaged.
References
- https://www.gisaid.org accessed 20 December 2021
- Hadfield J, Megill C, Bell SM et al. Nextstrain: real-time tracking of pathogen evolution. Bioinformatics. 2018; 34: 4121-4123.
- Rambaut A, Holmes EC, O’Toole Á et al. OGA dynamic nomenclature proposal for SARS-CoV-2 lineages to assist genomic epidemiology. Nat Microbiol. 2020; 5:1403-1407.
- O’Toole A, Scher E, Underwood A et al. Assignment of epidemiological lineages in an emerging pandemic using the pangolin tool (2021). Virus Evolution DOI:10.1093/ve/veab064
- https://www.who.int/en/activities/tracking-SARS-CoV-2-variants
- https://www.samrc.ac.za/news/tshwane-district-omicron-variant-patient-profile-early-features Accessed 20 December 2021
- Sandile C, Jackson L, Khadija K et al. SARS-CoV-2 Omicron has extensive but incomplete escape of Pfizer BNT162b2 elicited neutralization and requires ACE2 for infection. 2021 https://www.scribd.com/document/545376307/MEDRXIV-2021-267417v1-Sigal
- Wilhelm A, Widera M, Grikscheit K et al. Reduced neutralization of SARS-CoV-2 Omicron Variant by vaccine sera and monoclonal antibodies. medRxiv. 2021:2021.12.07.21267432
- Basile K, Rockett RJ, McPhie K et al. Improved neutralization of the SARS-CoV-2 Omicron variant after Pfizer-BioNTech BNT162b2 COVID-19 vaccine boosting. bioRxiv 2021.12.12.472252; doi: https://doi.org/10.1101/2021.12.12.472252
- Andrews N, Stowe J, Kirsebom F et al. Effectiveness of COVID-19 vaccines against the Omicron (B.1.1.529) variant of concern. https://khub.net/documents/135939561/430986542/Effectiveness+of+COVID-19+vaccines+against+Omicron+variant+of+concern.pdf/f423c9f4-91cb-0274-c8c5-70e8fad50074
- Barnard R, Davies NG, Pearson CAB et al. Modelling the potential consequences of the Omicron SARS-CoV-2 variant in England. 2021. https://cmmid.github.io/topics/covid19/reports/omicron_england/report_11_dec_2021.pdf
- Hogan AB, Wu SL, Doohan P et al. The value of vaccine booster doses to mitigate the global impact of the Omicron SARS-CoV-2 variant. Imperial College London (15-12-2021), doi: https://doi.org/10.25561/93034
- WHO Report September 2021 WHO COVID-19 Vaccines Research. Will emerging data allow increased reliance on vaccine immune responses for public health and regulatory decision-making? https://cdn.who.int/media/docs/default-source/blue-print/ who-op_3 sept2021_ v3.pdf?sfvrsn= a20d5d39_7.
- VanBlargan LA, Errico JM, Halfmann P et al. An infectious SARS-CoV-2 B.1.1.529 Omicron virus escapes neutralization by several therapeutic monoclonal antibodies. bioRxiv 2021.12.15.472828; doi: https://doi.org/10.1101/2021.12.15.472828
- Dougan M, Nirula A, Azizad M, et al. Bamlanivimab plus Etesevimab in Mild or Moderate Covid-19. N Engl J Med. 2021;385(15):1382-1392. doi:10.1056/NEJMoa2102685
- Regkirona SmPC: https://www.ema.europa.eu/en/documents/product-information/regkirona-epar-product-information_en.pdf Accessed 20 December 2021
- Weinreich DM, Sivapalasingam S, Norton T, et al. REGEN-COV antibody cocktail clinical outcomes study in covid-19 outpatients. N Engl J Med 2021; 385:e81 DOI: 10.1056/NEJMoa2108163
- Gupta A, Gonzalez-Rojas Y, Juarez E et al. Effect of the Neutralizing SARS-CoV-2 Antibody Sotrovimab in Preventing Progression of COVID-19: A Randomized Clinical Trial. medRxiv 2021.11.03.21265533; doi: https://doi.org/10.1101/2021.11.03.21265533
- Cohen MS, Nirula A, Mulligan MJ, et al. Effect of Bamlanivimab vs Placebo on Incidence of COVID-19 Among Residents and Staff of Skilled Nursing and Assisted Living Facilities: A Randomized Clinical Trial. JAMA. 2021;326(1):46-55. doi:10.1001/jama.2021.8828
- O’Brien MP, Forleo-Neto E, Musser BJ et al. Covid-19 Phase 3 Prevention Trial Team. Subcutaneous REGEN-COV Antibody Combination to Prevent Covid-19. N Engl J Med. 2021; 385:1184-1195. doi: 10.1056/NEJMoa2109682
- Evusheld US Healthcare Provider Fact Sheet https://www.fda.gov/media/154701/download Accessed 20 December 2021
- Biegel JH, Tomashek LE et al. Remdesivir for the treatment of Covid-19 – final report. N Engl J Med 2020; 383:1813-1826 DOI: 10.1056/NEJMoa2007764
- Hill J et al. Three-day course of remdesivir may help stave off COVID-19–related hospitalization and death: results of the phase III PINETREE study. ID Week 2021, abstract LB1
- Meeting Highlights CHMP Meeting 13-16 Dec 2021 https://www.ema.europa.eu/en/news/meeting-highlights-committee-medicinal-products-human-use-chmp-13-16-december-2021
- https://www.gilead.com/news-and-press/company-statements/gilead-statement-on-veklury-remdesivir-and-the-sars-cov-2-omicron-variant
- Bernal AJ, Gomes da Silva MM, Musungaie DB et al. Molnupiravir for Oral Treatment of Covid-19 in Nonhospitalized Patients. N Engl J Med. DOI: 10.1056/NEJMoa2116044.
- Owen DR, Allerton CMN, Anderson AS et al. An Oral SARS-CoV-2 Mpro Inhibitor Clinical Candidate for the Treatment of COVID-19. medRxiv doi: https://doi.org/10.1101/2021.07.28.21261232
- EMA Advice Release Dec 2021: https://www.ema.europa.eu/en/news/ema-issues-advice-use-paxlovid-pf-07321332-ritonavir-treatment-covid-19-rolling-review-starts
- Malsy J, Veletzky L, Heide J et al. Sustained response after remdesivir and convalescent plasma therapy in a B-cell depleted patient with protracted COVID-19. Clin Infect Dis an Off Publ Infect Dis Soc Am. 2020; 6:ciaa1637.
- Helleberg M, Niemann CU, Moestrup KS et al. Persistent COVID-19 in an Immunocompromised Patient Temporarily Responsive to Two Courses of Remdesivir Therapy. J Infect Dis. 2020; 222: 1103–7.
- Gandhi S, Klein J, Robertson A et al. De novo emergence of a remdesivir resistance mutation during treatment of persistent SARS-CoV-2 infection in an immunocompromised patient: A case report. medRxiv. 2021 Jan 1;2021.11.08.21266069.
- NERVTAG Briefing December 2021 https://assets.publishing.service.gov.uk/government /uploads/system/uploads/attachment_data/file/1039516/S1430_NERVTAG_Antiviral_drug_resistance_and_use_of_Direct_Acting_Antiviral_Drugs_.pdf
- Ison MG, Hayden FG, Hay AJ et al. Influenza polymerase inhibitor resistance: Assessment of the current state of the art – A report of the isirv Antiviral group. Antiviral Res. 2021 Oct;194:105158. doi: 10.1016/j.antiviral.2021.105158. Epub 2021 Aug 4. PMID: 34363859.


